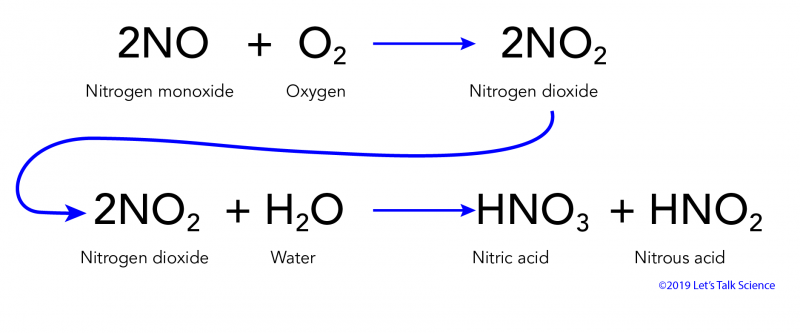
Acid rain is caused by emissions of sulfur dioxide and nitrogen oxide which react with the water molecules in the atmosphere to produce acids.
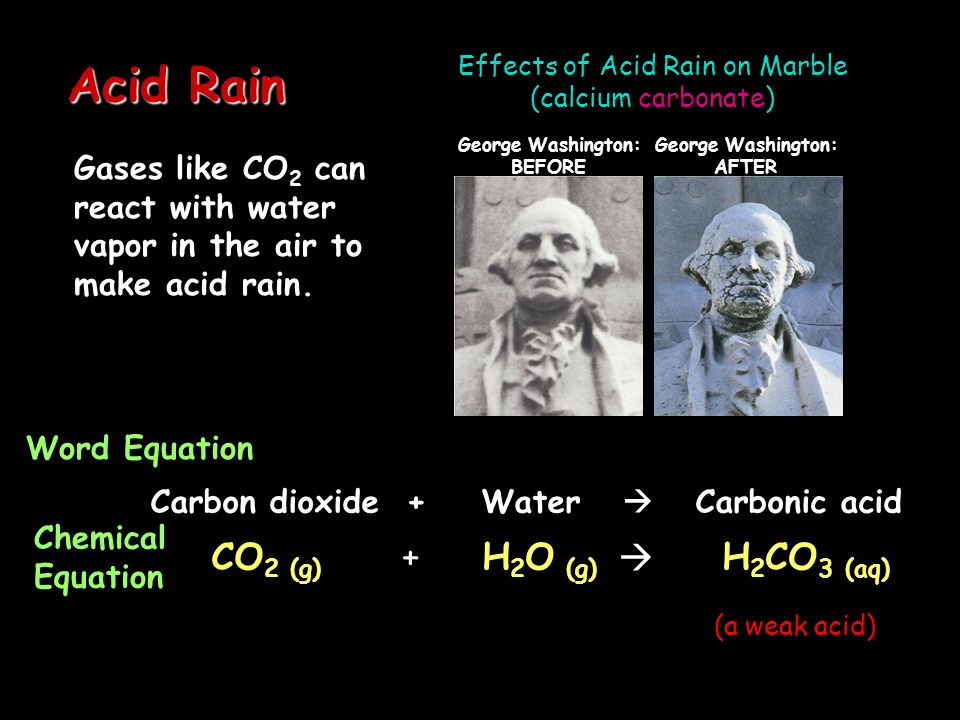
Chemical formula for acid rain and marble.
Although these are recognized as highly durable materials buildings and outdoor monuments made of marble and limestone are now being gradually eroded away by acid rain.
Figure 4 18 acid rain damage to a statue of george washington.
It happens on the surface of the marble or limestone and the fine details on the facade of the structures made with them are being lost.
Acid rain acid rain chemistry of acid deposition.
When sulfurous sulfuric and nitric acids in polluted air react with the calcite in marble and limestone the calcite dissolves.
What effect does acid rain have on marble.
Acid rain is a popular expression for the more scientific term acid deposition which refers to the many ways in which acidity can move from the atmosphere to earth s surface.
Both marble and limestone consist of caco 3 which reacts with acid rain in an acid base reaction to produce caso 4.
Acid rain contains sulfuric acid which reacts with calcium carbonate to create aqueous ions.
How does acid precipitation affect marble and limestone buildings.
Stone surface material may be lost all over or only in spots that are more reactive.
Acid rain is a rain or any other form of precipitation that is unusually acidic meaning that it has elevated levels of hydrogen ions low ph it can have harmful effects on plants aquatic animals and infrastructure.
Acid rain damage to a statue of george washington.
Acid deposition includes acidic rain as well as other forms of acidic wet deposition such as snow sleet hail and fog or cloud water.
Because caso 4 is somewhat soluble in water significant damage to the structure can result.
Both marble and limestone consist of caco 3 which reacts with acid rain in an acid base reaction to produce caso 4.
Acid precipitation affects stone primarily in two ways.
How does this happen.
In exposed areas of buildings and statues we see roughened surfaces removal of material and loss of carved details.
The marble will begin breaking down as a result of the acid.
The biological effects of acid rain are more complex.
However since the onset of acid rain these materials are at risk from erosion.
A chemical reaction equation 9 between calcium carbonate and sulfuric acid the primary acid component of acid rain results in the dissolution of caco.
Students know how to use the ph scale to characterize acid and base solutions.
When sulfurous sulfuric and nitric acids in polluted air and rain react with the calcite in marble and limestone the calcite dissolves.
While marble is a strong material there are some conditions that it cannot stand.
Because caso 4 is somewhat soluble in water significant damage to the structure can result.
Students know how to calculate ph from hydrogen ion concentration.